Chemical engineering

Towards the end of my junior year I started to participate in an after school program held at an organization called Inspiring Minds. The program was all about chemical engineering. We learned about all different kinds of jobs that are out there in this field and we learned about all sorts of products that chemical engineers create. I sadly joined the program a little late so I missed a portion of the classes, but I still showed up and participated every Wednesday. At the end of the program I wound up getting an award for being the most engaged student in the class.
Homemade bouncy balls
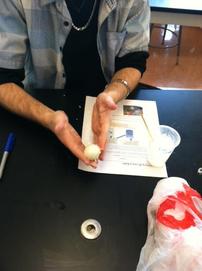
This was the first class I joined in on. We made bouncy balls out of borax, cornstarch, and glue. The bouncy balls we made were made from a polymer. Polymers are molecules made up of repeating chemical units. Glue contains the polymer polyvinyl acetate, which cross links to itself when reacted with borax. This is what makes the bouncy ball retain its form. I was the only one in the class who was able to make an actual bouncy ball. Everyone else's ball couldn't retain its sphere shape, or would just stick to the table when bounced...I was very proud of my bouncy ball.
Running on oobleck
Oobleck is a non-newtonian fluid. It acts like a liquid when being poured, but acts like a solid when a force is acting on it. You can grab it, and it will ooze out of your hand. Make enough of it and you can actually walk on it (which is exactly what we did). Oobleck is made when you mix cornstarch and water together. The goal is to get a consistency where the Ooblech reaches a state that is a liquid, yet solid.

